2025
Presently (Q3) 2025, already seven insect Novel Food ingredients have been authorised, following the Commission Implementing Regulation of 20 January, authorising the placing on the market of UV-treated powder of whole yellow mealwom (Tenebrio molitor larvae) as a novel food ingredient, corresponding to the third yellow mealworm authorised NF product.
In 2025, 10 EFSA positive Safety opinions have been published on insects as Novel Food ingredients, following the EFSA opinion on yellow mealworm (Tenebrio molitor) on the 16th of January.
2024
By the end of 2024, six insect Novel Food ingredients have been authorised, while nine positive EFSA opinions have been published on insect Novel Food ingredients.
On the 16th of December 2024, EFSA published its 9th positive opinion on insects as a Novel food ingredient, covering frozen, dried and powder forms of house crickets (Acheta domesticus) pursuant to Regulation (EU) 2015/2283.
On 29 July 2024, EFSA published its 8th positive opinion for edible insects as Novel Food, on house cricket powder (Acheta domesticus).
On 1 June 2023, EFSA published its 7th positive opinion for edible insects as Novel Food, corresponding to the one on UV-treated powder of whole yellow mealworm (Tenebrio monitor).
The respective European Commission Implementing Regulations authorising their commercialisation are expected to be published soon.
2023
Following EFSA’s 5th opinion from May 2022, in 2023 the European insect sector also welcomed the Commission’s Implementing Regulation authorising partially defatted whole house cricket (Acheta domesticus) as Novel food, corresponding to the second authorised Novel Food ingredient from house cricket . The authorisation came into effect on 24 January 2023.
2022
At the beginning of 2022, the European Commission published the second Implementing Regulation authorising the commercialisation of the Novel Food ingredient from frozen, dried and powder yellow mealworm (Tenebrio Molitor), and the first authorisation of dried, powder and frozen house cricket (Acheta domesticus), following the 3rd and 4th EFSA safety opinions from August 2021. The authorisations entered into force on 1st and 3rd of March 2022, respectively.
In May 2022, the Parma-based Agency (EFSA) published the 5th opinion on edible insects, namely on the partially defatted house cricket (Acheta domesticus).
On 4 July 2022, EFSA published its 6th opinion on edible insects, namely on the safety of frozen and freeze-dried formulations of the lesser mealworm (Alphitobius diaperinus larva). This authorisation is the second for this species and follows the EFSA opinion published in May 2022.
2021
Following the 1st EFSA opinion covering insect food ingredients as Novel Food (January 2021), dried yellow mealworm (Tenebrio molitor) has been authorised at the EU level as the first insect Novel food ingredient following the enter into force of the Commission Implementing Regulation authorising its commercialisation, entering into force in June 2021.
Following EFSA’s 2nd positive opinion (July 2021), the European Commission authorised dried and frozen migratory locust (Locusta migratoria) as a Novel Food ingredient, following the entering into force of the Commission Implementing Regulation authorising its commercialisation at the EU level, which entered into force on 3 December 2021.
Learn more:
Also read more on how Novel Food ingredients are authorised to commercialised at the EU level and on the different types of Novel Food applications:


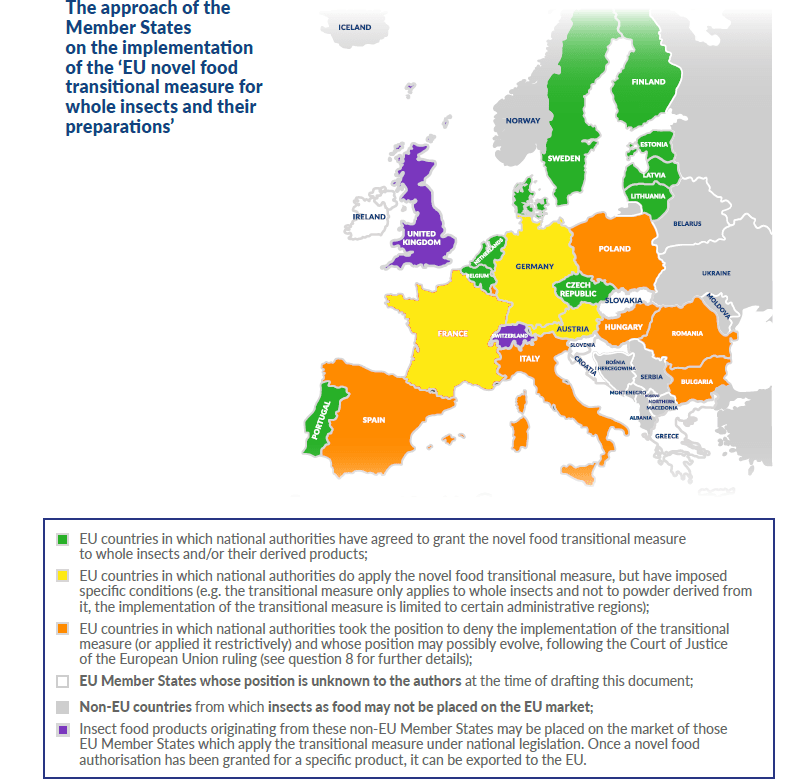

 Summary overview of the respective scopes of Regulation (EC) No 258/97 and Regulation (EU) 2015/2283 in respect of whole insects, parts of whole insects and insect-based preparations
Summary overview of the respective scopes of Regulation (EC) No 258/97 and Regulation (EU) 2015/2283 in respect of whole insects, parts of whole insects and insect-based preparations